#4. Are your clothes actually warm?
Common misconceptions about heat and cold
As winter is approaching in the northern hemisphere, the topic of keeping yourself warm is becoming relevant again. Unless you’re moving to some warm location elsewhere on the planet Earth, your wardrobe is most likely going to change. It will include more “warm” clothes, as we call them.
But what does “warm” actually mean? Let’s look at it more deeply from a scientific standpoint.
🌡️ Temperature vs Warmth
First of all I want to define the terms that I’m using, to avoid potential confusion.
🌡️ Temperature – is an objective property of matter that can be precisely measured by an instrument. It can be expressed in various units of measure, like ºC (Celsius), ºF (Fahrenheit) or ºK (Kelvin), which are simply different conventions that certain groups of people have agreed upon. Yet the key aspect is that as long as properly functioning instruments are used to measure it and the relevant unit conversions are applied – every measurement will show the same value, no matter who had done it. Temperature is objective.
🥵 Warmth – is a subjective measure of how warm (or cold) something feels to a specific person, where the objective temperature of the substance is only one of the aspects contributing to that feeling. I’ve touched upon this in my introductory article, mentioning how cold water felt burning hot to my freezing hands during the Ukrainian winter. Similarly, Italians are usually surprised by how comfortable British people look wearing summer clothes in winter, which would feel unbearable in Italy. Same objective temperature is perceived as different degrees of warmth (or cold) by different people.
I don’t want to go into the deeply personal aspects of temperature perception, which have more to do with biology and psychology than physics. Instead, I will focus on the technical aspects, which are more universal and work the same regardless of personal variations.
🥵 Humans as walking heaters
We, humans, are warm-blooded mammals with the healthy body temperature of around 37ºC, which we need to maintain in order to keep all the internal biochemical processes functioning properly. Most of the time the temperature of our environment is lower than that, so the body constantly consumes energy to keep itself at this higher temperature despite the colder environment. Normally this extra heat is the natural by-product of our metabolism, so we don’t even notice it.
💡 This is why you have to eat every day to stay alive, even when simply lying in bed and doing nothing. In a way we are natural heaters that run on food instead of electricity.
When the outside temperature becomes so low that the normal metabolism can’t produce enough heat, additional mechanisms kick in, like contraction of the outer blood vessels and shivering. These are much harder to miss – a clear signal to our conscious mind that we should find a more sustainable and comfortable way to keep our body at its design temperature.
↔️ Heat transfer rules it all
We perceive external temperature through thermoreceptors1 embedded in our skin, which detect the direction and intensity of heat transfer between the skin and the outside world. Unlike a thermometer, which measures the objective absolute temperature, our skin can only feel how much something is warmer or colder than itself. Therefore, our subjective feeling changes with the temperature of our own skin.
This suggests that you should feel the coldness of the surrounding air pretty much all the time, since even in summer the air temperature rarely goes above 37ºC. First of all, your skin is colder than your inner body, since it stays in direct contact with the air that cools it down. Therefore, its baseline temperature is somewhere closer to 30ºC. But the main catch is in the intensity of the heat transfer between your skin and the air.
Even though room-temperature air at 25ºC is definitely colder than your skin, you’d feel perfectly comfortable in it, because the rate at which it is taking away your heat typically isn’t high enough to feel it as cold. I say “typically”, because there are nuances…
🌈 Thermal aura
When you are standing still surrounded by cold air, your body is losing heat primarily through 2 mechanisms:
🔆 radiation (~60%) – the one captured by infrared thermal cameras, emitted from your body like rays from a lightbulb (typical wavelength: 0.012 mm);
💨 convection (~40%) – substance absorbing heat through direct contact with your skin (typical air conductivity: 24 mW/(m·K)).
💡 Don’t confuse the “heat radiation” with “ionising radiation” that comes from radioactive materials, X-rays or UV-rays. These are completely different phenomena that have very little in common. I will talk about it more deeply in another blog post.
The key distinction between these two mechanisms is that infrared radiation leaves your body and travels until it is absorbed or reflected by something (air is essentially transparent for infrared rays). Convection, on the other hand, only works through direct contact with your skin.


Since air has a fairly low thermal conductivity, convection creates a layer of warmer air around your skin, which looks like a slowly-evaporating aura. This is why your skin doesn’t really feel the actual temperature of the air, but a warmer version of it. This also explains why you feel much more comfortable in 20ºC air than in 20ºC water, which has a much higher heat conductivity, taking away your heat way faster.
💨 Gone with the wind
We all know that the wind makes us feel colder, and before becoming a physicist I assumed that this is simply because the wind makes the air colder, without ever questioning the mechanism of it. But at some point, after I’ve learnt about the basic principles of thermodynamics, it struck me that just the air being in motion cannot lower its own temperature. And you can easily verify it by placing a thermometer in front of a fan – the air blowing at it will not change its readings at all.
It only starts making sense once you replace the thermometer with something that is producing heat itself, like your body covered with the “thermal aura“. As the wind blows at you, it strips away that heated air, replacing it with the new colder air, which is ready to absorb more heat from your skin through convection. The same thing happens if the air stands still and you’re moving instead – like walking or riding on an electric scooter.
So technically the wind doesn’t arbitrarily cool you down. It just makes you feel the actual air temperature instead of the one heated by your body. If you think this is a useless change of words saying exactly the same thing, here are a few logical consequences that might not be obvious if you consider wind as a simple “cooler“:
the stronger is the wind, the colder it feels, as you’ve surely experienced yourself, but only up to a certain point. Eventually new colder air will be incoming faster than your skin can give away its heat through convection, meaning that any further increase of wind speed will not make it feel any colder. In other words, if your fan had extra speed settings of 4-…-10 in addition to the usual 1-2-3, the 8-9-10 would all feel the same. This resembles the terminal velocity of raindrops that don’t kill you, which I’ve mentioned in my earlier article.
the wind only cools you down because the surrounding air is colder than you. You would feel no effect in a room at 37ºC, while at 40ºC the wind would actually heat you up instead of cooling you down.
💡 I have to mention that in practice even the warm wind can still cool you down through evaporation of sweat. This is another temperature-regulation mechanism in our body that has plenty of its own interesting aspects closely tied to humidity, which are worth a dedicated publication. In cold weather it’s less relevant though.
🧥 Clothes as thermal insulators
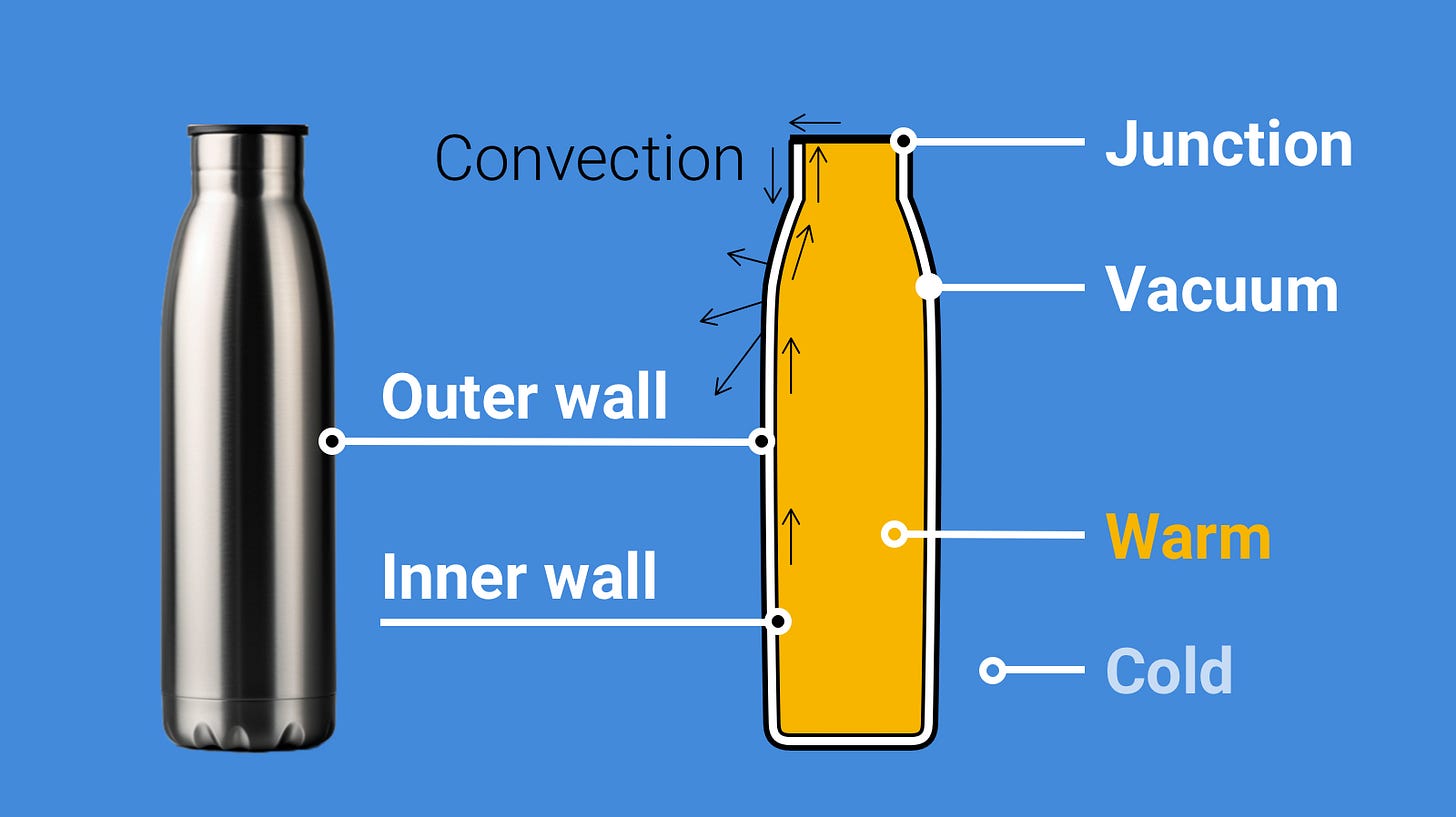
The main objective of warm clothes is to reduce the heat loss of your body as much as possible. Among everyday objects you can find the ultimate thermal insulation in a thermal flask (aka thermos).
Its key element is the vacuum between two walls, preventing convection. Thus, the only way for the heat to leave the flask is through the tiny junction between the two walls at the top, where heat can pass from the inner wall to the outer one and then to the outside air. Achieving vacuum in everyday clothes is pretty much impossible, so our aim is to get as close as possible to the thermos effect using more practical and wearable structures – fabrics.
👕 T-shirt anatomy
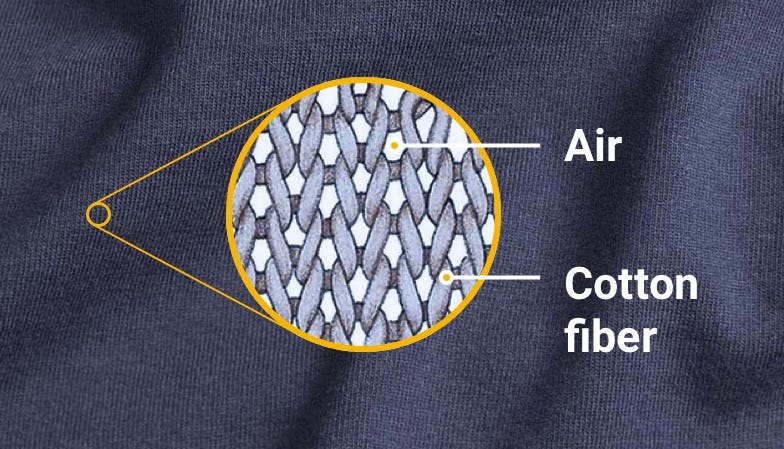
Let’s zoom into a Jersey fabric – the most common material in T-shirts. Essentially it’s a repeating pattern of fibers with pockets of air in-between.
The air has low heat conductivity (~26 mW/(m∙K))2, whereas fibers have high conductivity (~40 mW/(m∙K))3. So in simple terms, your body heats the fibers, which hold the warmed air inside, making it harder to replace by the external cold air. In analogy to the thermal flask – fibers act like the junction between two walls and air acts as a vacuum between them.
Translating these concepts to a fabric, its thermal performance depends on 2 closely intertwined aspects:
material composition of the individual fibers, which defines how well the heat propagates from your body to the air;
weave or knit, which defines the pattern of how the fibers are put together into the actual fabric and, respectively, how much air it can hold.
Typically you can find materials that are purely natural (cotton, wool, silk, linen) or synthetic (acrylic, polyester, nylon) or a blend of the two. Silk or polyester feel cold to the touch, because their fibers are very good heat conductors, whereas wool or acrylic are at the opposite side of the spectrum – warm and bad heat conductors.
Obviously, the thicker is the fabric, the longer is the path and the larger is the number of air pockets that the heat needs to go through → better thermal insulation ✅. But also, thicker fabric makes it more bulky, heavier and harder to style 🛑. So unless you’re happy to wear a heavy and thick knitted wool sweater, you can find a smarter way to achieve a similar level of thermal insulation.
🥪 Sandwich style
As I’ve mentioned above, it’s actually the air that has the lowest thermal conductivity – not the material of the fabric itself. So we can use that feature to our advantage by adding air to the equation. For example, if we split a single layer of fabric in two, we’ll end up with a thin layer of air in-between that will act as extra insulation, which is practically weightless. And the more air you can hold – the better, which is why they should not be perfectly smooth, but have some structure to them, like fleece, corduroy or deep knit.
Some of these fabrics might not feel very nice to touch, so you’d probably want to add a thin layer of softer fabric underneath. It doesn’t have to be dense – just enough to prevent the contact with your skin. As always, air between the fibers would only help.
Finally, you want to prevent the warm air from being blown away easily, which is why it’s important to have an outside wind-proof layer. That’s where synthetic materials are particularly effective, like tightly-woven nylon or polyester, which are more lightweight than natural leather or pressed wool felt.
So to summarise, a perfect sandwich of clothes would look something like this ⬇️ depending on how warm you want it to feel. Extra warmth requires an extra-air-holding material, such as goose down or fur. In some warm jackets you can even find a layer of perforated aluminium foil to reflect infra-red radiation back into your body.
💦 Beware of Sweat
When pursuing your perfect combination of warm clothes, it’s important to remember – don’t exaggerate. Making yourself too warm will activate your natural cooling mechanism – sweating. As you sweat, the fibers of your clothes get wet, significantly increasing their heat conductivity.
This is another reason why having multiple layers is beneficial. If you do sweat, the water will mostly absorb into the 1st layer, and much less into the 2nd one, which is separated by the air gap. This will limit the damage to your heat insulation and give you the opportunity to take that 1st layer off, keeping the rest of the clothes intact.
Finally, having multiple layers gives you more flexibility in regulating how warm you want to feel, simply by taking off one layer or the other. This is particularly useful if you’ve entered a warm room/bus/train or about to do a physical activity, like walking fast or carrying a heavy bag.
Without adjusting your level of thermal insulation, even a few minutes of such disbalance may cause enough sweat to turn your warm clothes into a cooler. Even though it feels good in the process, as soon as you get back to the previous conditions – cold air with no physical activity – you’ll start freezing. And restoring the original level of insulation by evaporating all that sweat will take a long time – often enough to catch cold 🥶
This is where preventive action is key❗️
🎬 Epilogue
Understanding how the heating technically works allows you to choose clothes wisely and achieve the best result for the given environment, availability of materials, budget, etc.
Just don’t forget that it’s not the clothes that make you warm – it’s your body. Warm clothes is just one of the methods to make you feel warm. You can also produce more heat, by walking faster or jumping on the spot, burning more energy as a result. Or you could change your diet to add a healthy layer of fat under your skin – a built-in thermal insulator4.
Finally, depending on the distribution of fat, blood-circulation and hair across your body, you might have significant heat loss through other parts of your body, like feet, hands or head. So if you can’t or don’t want to insulate every single part of your body, focus on the ones that have the maximum value of temperature times area.
We’re all different, so do what fits you best. Just approch it keeping the science in mind.
Thermal conductivity of air, The Engineering Toolbox
Thermal conductivity assessment of cotton fibers from apparel recycling for building insulation, Energy and Buildings (324) 1 December 2024
Do Fat People Stay Warmer in Cold Weather Than Thin People?, The Weather Channel